|
HEAVY
METALS
ABOUT -
CONTACTS - FOUNDATION -
HOME - A-Z INDEX
HEAVY
METAL - Plastic is like a
sponge to many metals in our oceans.
NEWS DEEPLY APRIL 3 2018 - HEAVY METAL,
THE NEW DANGER POSED BY OCEAN PLASTIC
Plastics entering coastal waters both absorb and release cadmium, lead and other toxic metals. Scientists are now trying to determine the impact of metal-contaminated plastic on marine life and ocean ecosystems.
We know that plastic waste is overwhelming the ocean, sea life is dying from ingesting it and some even ends up in seafood. But scientists also now worry that plastic trash is coming with a side helping of toxic metals that latch onto plastic surfaces and enter the marine environment and food chain – and eventually, what people eat.
Metals, such as cadmium and lead, are often used in manufacturing plastic and over time can enter coastal waters. Once floating in the ocean or discarded on a beach and washed by the tides, plastics can also attract and concentrate a variety of metals already present in the environment that attach themselves, or “sorb,” to the surface. In both cases, the worry is that these metals – often toxic ones such as cadmium that are health concerns for both wildlife and humans – can contaminate waters or harm wildlife that ingest plastics, especially those that live in intertidal zones near sources of plastic pollution.
Researchers, however, are only just starting to understand how metal-tainted plastics interact in coastal environments, said Leah Bendell, professor of marine ecology and ecotoxicology at Simon Fraser University in British Columbia.
Bendell led one recent study, published in February in the journal PLOS ONE, that examined how four metals – cadmium, lead, zinc and copper – both attach onto and are released from plastics found on Canada’s beaches. She said her results show how a whole host of metals can enter the marine food chain or coastal waters.
“Not only were these plastics serving as a way of metal getting into these lower trophic levels, but also they were a source of the metal into the water column and they can be acutely toxic,” said Bendell. “It was a little bit of an eye-opener to the multifaceted role the plastics played.”
For the study, Bendell’s graduate student, Bertrand Munier, picked up every bit of plastic waste from transects on nine Vancouver-area beaches, gathering 144 unique plastic items, mostly food packaging and takeout containers. They sorted the plastics into 11 types and then used a weak acid to extract and separate the four metals – this kind of analysis is often used to estimate levels of toxins that could enter the tissues of wildlife if eaten. As a point of comparison, they also did the same for newly manufactured plastic samples. The goal was to distinguish metals that came from the plastic itself and those that had sorbed to the surface of the beach debris from the environment.
Of the collected items, five samples released what the study said were “extreme” high levels of metals – including a plastic tampon applicator tested for high levels of zinc – and all had at least trace amounts of the four metals tested. Different kinds of plastic also released different levels of metals. For example, PVC, the most commonly found plastic, had high levels of lead and copper attached to its surface. The comparison of the new and debris plastic also showed how some of the chemicals used in plastic production may release over time – including cadmium, which is used to make plastic rigid and resistant to UV light. The researchers found that new
PVC releases zinc and cadmium.
A previous study examining metals sorbing onto plastics have found that the age of the material also matters. Chelsea Rochman, an assistant professor at the University of Toronto’s department of ecology and evolutionary biology, led a study when she was at San Diego State University in which her team dropped mesh bags of various kinds of plastic pellets into three areas around San Diego Bay in California. They measured how much aluminum, chromium, manganese, iron, cobalt, nickel, zinc, cadmium and lead from the environment sorbed onto their samples.
The year-long study, also published in PLOS ONE, found that metal levels increased the longer the plastic samples were in the water. That’s probably because surface area increases as the plastics degrade over time and biofilms form, Rochman said.
Biofilms are collections of unattached microorganisms that put down roots on surfaces and can act as a surface for metals to latch onto. Fungi are a type of biofilm, as are bacteria. “Basically, over time there’s more space for these metals to bind to,” said Rochman.
There’s still a lot scientists don’t know. For example, it’s unclear how big a role biofilms play in the concentration of metals on plastics and the ultimate effects of the metals on wildlife that ingest plastics. It’s possible, for example, they may digest the biofilm, metals or chemicals – even if they ultimately expel the plastic itself. “If the metals are bound on the biofilm, the question is are they even more bioavailable than we think?” asked Rochman.
The presence of a toxic metals-saturated biofilm on plastics could be both an ecological and human health problem, said Bendell. The bacterial growth on the biofilm could potentially pick up pathogens in and around coastal areas. And as these plastics break down into smaller and smaller pieces, they’re more easily ingested by marine life, and now it looks like they’re bringing dangerous metals along for the ride. While the studies were conducted in North America, the environmental risks may be far greater in regions like Southeast Asia that lack waste management infrastructure and where more plastic pollution makes its way to the coast.
The actual risk of metals associated with plastics to human health is unknown, said Bendell. But as plastic pollution grows, it’s concerning to scientists like Bendell. “We need to change from thinking everything can be thrown away to you are accountable and responsible for every piece of
plastic that comes into your house,” she said.
BEACHES - You
can't see it in the ocean, but you know it is there because of
the tides washing the stuff up
on our beaches. It also works in reverse if you throw litter
away while on holiday.
THE
INDEPENDENT MAY
2016 - MPS TOLD PLASTIC TOXIC ONCE IN THE
ENVIRONMENT
Microplastics – which accumulate chemicals that disrupt human hormones – are getting into seafood like North Sea
mussels and
Atlantic
oysters.
Plastic should be considered “toxic” if it gets into the natural environment because of its ability to attract poisonous chemicals “like a magnet”, according to an international campaign group.
In a submission to a parliamentary inquiry into the environmental impact of microplastics, the Plastic Oceans Foundation warned that tiny pieces of plastic laden with chemicals were finding their way through the gut into the flesh of seafood and, from there, into the human food chain.
Microplastic comes from a range of sources, such as microbeads deliberately added to soap as an exfoliant and the 1,900 tiny fibres that can be produced each time a single piece of synthetic clothing is washed. Larger pieces of plastic also break down over time into microscopic pieces.
The House of Commons’ Environmental Audit Committee is due to hold the first hearing of its inquiry into the effects of microplastics on Monday, 9 May.
In its written submission to the committee, the Plastic Oceans Foundation said: “Evidence clearly shows that plastics contain or are formulated with chemicals that are known to be toxic, such as phthalates, and bisphenol A, and which cause a broad range of effects including critical diseases and health conditions.
“Plastic also attracts chemicals ‘like a magnet’ the moment it enters the ocean, as well as causing physical harm to marine life.”
The chemicals accumulate in “body tissue, particularly fat and muscle tissue, and biomagnifies in the marine food chain” and then enters human food supply, the campaign group added. This can affect the human endocrine system, which produces hormones involved in growth, the metabolism, sexual development and function.
“These toxic substances... through a variety of toxicological mechanisms including endocrine disruption can accumulate into the human food chain through consumption of fish and cause health effects in the human population,” it said.
“Plastics should therefore be considered to be toxic when released to the environment.”
The charity plans to highlight the problem in a documentary film called A Plastic Ocean, which is due to be released later this year, featuring scientists and commentators including
Sir David
Attenborough.
In its submission to the committee, the Department for Environment, Food and Rural Affairs (Defra) noted an international scientific review of research into the health effects of microplastics had found “a large degree of uncertainty surrounding this issue”.
However it added: “Several studies show that microplastics are present in sea food sold for human consumption, including mussels in North Sea
mussel farms and
oysters from the Atlantic.
“The presence of marine microplastics in seafood could pose a threat to food safety. However, due to the complexity of estimating microplastic toxicity, estimations of the potential risks for human health posed by microplastics in foodstuffs is not yet possible.”
Defra said no scientist had studied whether microplastics eaten by
humans could get into the tissues of the body.
“Although the gut may be an important barrier, there is a possibility that very small particles such as nanoplastics could penetrate gut tissues.
“Experiments in rats have showed that polystyrene microspheres of 50-100nm (1nm = 1 millionth of a mm) can be absorbed into the body through the gut and transported to the liver and spleen.”
But it concluded that even for people who eat a lot of seafood “dietary exposure to microplastic particles is likely to be relatively low compared with inhalation of microplastics”.
The British Plastics Federation and PlasticsEurope wrote that "many" cosmetic firms had voluntarily agreed to remove microbeads from their products.
But their focus was on the need to teach people to "act responsibly with their waste" and suggested there should be more "community-led litter picks".
"The public need to be aware of the consequences of littering and inappropriate disposal of waste," they said.
"Education needs to take place within schools particularly ensuring secondary school children continue in the good habits children learn at primary
school. However, education of the wider public is also needed as a report from Keep Britain Tidy found 62 per cent of people in England drop litter (28 per cent admit to it) meaning the problem extends beyond young people."
They also called for "adequate litter bins particularly on beaches".
"Once bins are full the public will put bags of waste next to bins often thinking this is the right things to do," the two plastic industry bodies wrote.
"These bags may be blown or broken open by wildlife scavenging for food and it is important this waste is removed as part of the bin emptying process."
By Ian Johston Environment Correspondent

CARRIER -
This is
just a small sample of the plastic packaging that you will
find in retails stores all over the world (and some fishing
rope). A good proportion
of this packaging - around 8 millions tons a year, will end up
in our oceans, in the gut of the fish we eat, in the stomachs
of seabirds and in the intestines of whales and other marine
mammals, taking with it POPs in greater concentrations that will end up in the food you
eat. Copyright photograph © 22-7-17 Cleaner
Ocean Foundation Ltd, all rights
reserved.
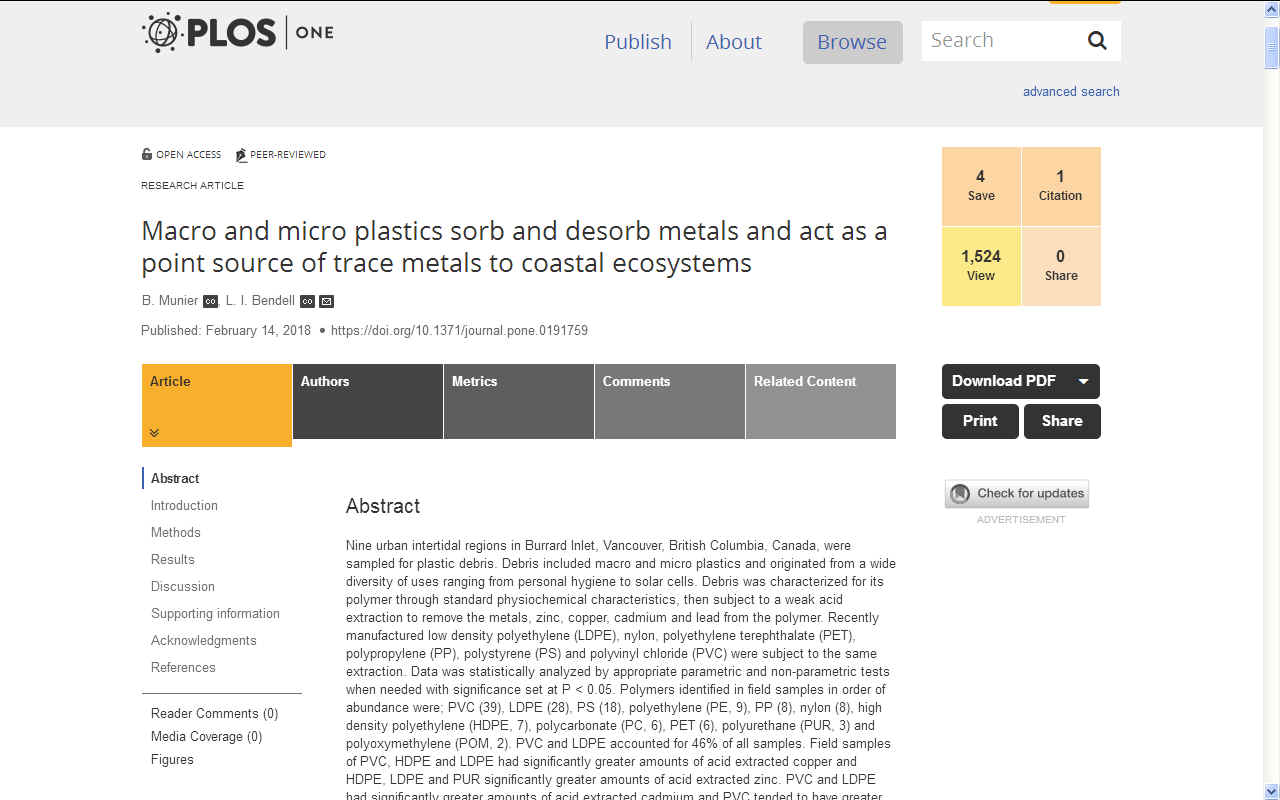
PLOS.ORG - PhytoNine urban intertidal regions in Burrard Inlet, Vancouver, British Columbia, Canada, were sampled for plastic debris. Debris included macro and micro plastics and originated from a wide diversity of uses ranging from personal hygiene to solar cells. Debris was characterized for its polymer through standard physiochemical characteristics, then subject to a weak acid extraction to remove the metals, zinc, copper, cadmium and lead from the polymer. Recently manufactured low density polyethylene (LDPE), nylon, polyethylene terephthalate (PET), polypropylene (PP), polystyrene (PS) and polyvinyl chloride (PVC) were subject to the same extraction. Data was statistically analyzed by appropriate parametric and non-parametric tests when needed with significance set at P < 0.05. Polymers identified in field samples in order of abundance were; PVC (39), LDPE (28), PS (18), polyethylene (PE, 9), PP (8), nylon (8), high density polyethylene (HDPE, 7), polycarbonate (PC, 6), PET (6), polyurethane (PUR, 3) and polyoxymethylene (POM, 2). PVC and LDPE accounted for 46% of all samples. Field samples of PVC, HDPE and LDPE had significantly greater amounts of acid extracted copper and HDPE, LDPE and PUR significantly greater amounts of acid extracted zinc. PVC and LDPE had significantly greater amounts of acid extracted cadmium and PVC tended to have greater levels of acid extracted lead, significantly so for HDPE. Five of the collected items demonstrated extreme levels of acid extracted metal; greatest concentrations were 188, 6667, 698,000 and 930 μgg-1 of copper, zinc, lead and cadmium respectively recovered from an unidentified object comprised of PVC. Comparison of recently manufactured versus field samples indicated that recently manufactured samples had significantly greater amounts of acid extracted cadmium and zinc and field samples significantly greater amounts of acid extracted copper and lead which was primarily attributed to metal extracted from field samples of PVC. Plastic debris will affect metals within coastal ecosystems by; 1) providing a sorption site (copper and lead), notably for PVC 2) desorption from the plastic i.e., the “inherent” load (cadmium and zinc) and 3) serving as a point source of acute trace metal exposure to coastal ecosystems. All three mechanisms will put coastal ecosystems at risk to the toxic effects of these metals.
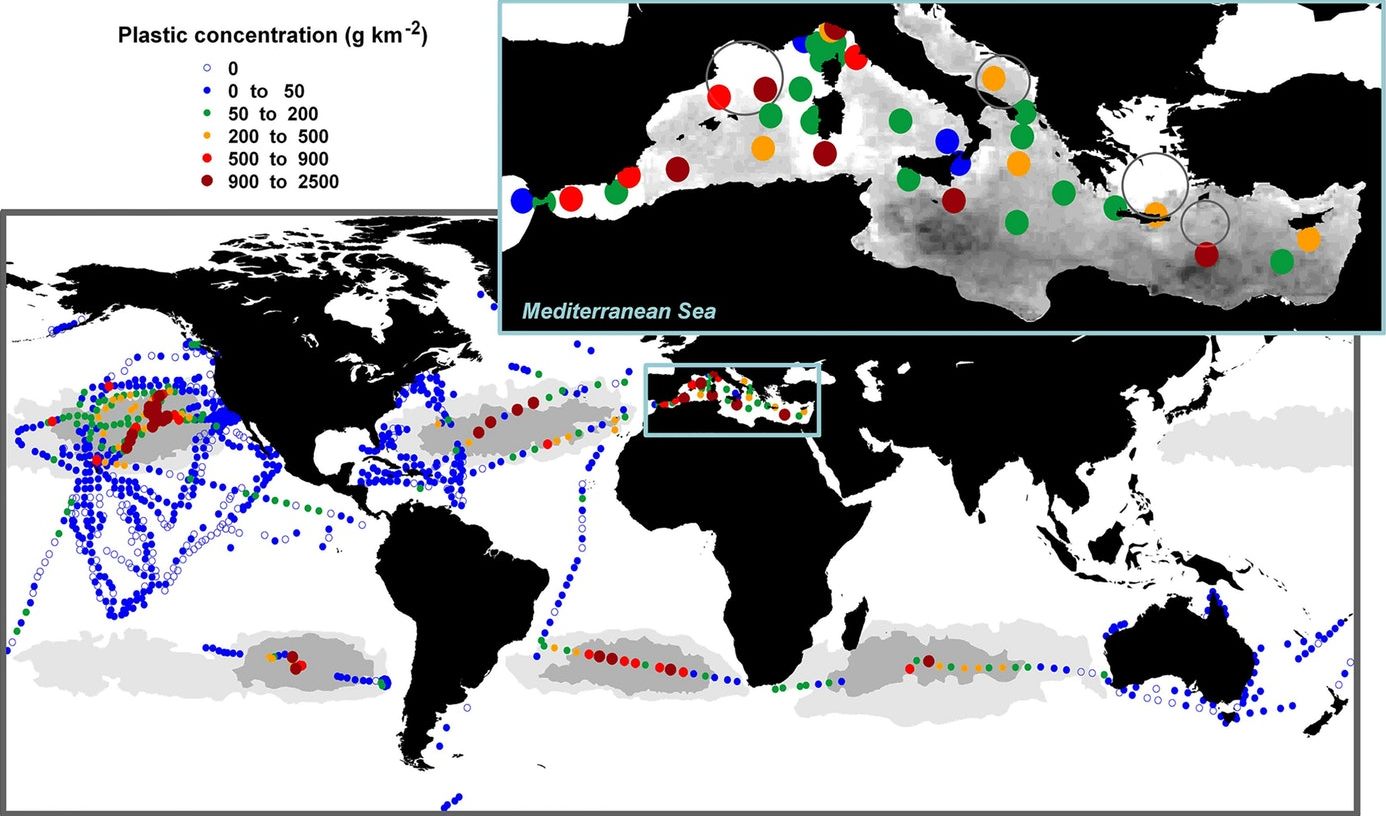
SATELLITES - European
Space Agency picture of plastic concentrations in our oceans.
MERCURY
POISONING
By
way of example, though mercury is only present in small amounts in seawater, it is absorbed by algae (generally as methylmercury). It is efficiently absorbed, but only very slowly excreted by organisms. Bioaccumulation and bioconcentration result in buildup in the adipose tissue of successive trophic levels: zooplankton, small nekton, larger fish, etc. Anything which eats these fish also consumes the higher level of mercury the
fish have accumulated. This process explains why predatory fish such as swordfish and sharks or birds like osprey and eagles have higher concentrations of mercury in their tissue than could be accounted for by direct exposure alone. For example, herring contains mercury at approximately 0.01 parts per million (ppm) and shark contains mercury at greater than 1
ppm.
PHYTOPLANKTON - The
basis of all life in the ocean is the humble phytoplankton,
the beginning of the ocean food chain, where photosynthesis
turns light energy into bio energy. On land plants turn light
energy into bio energy using the same process.
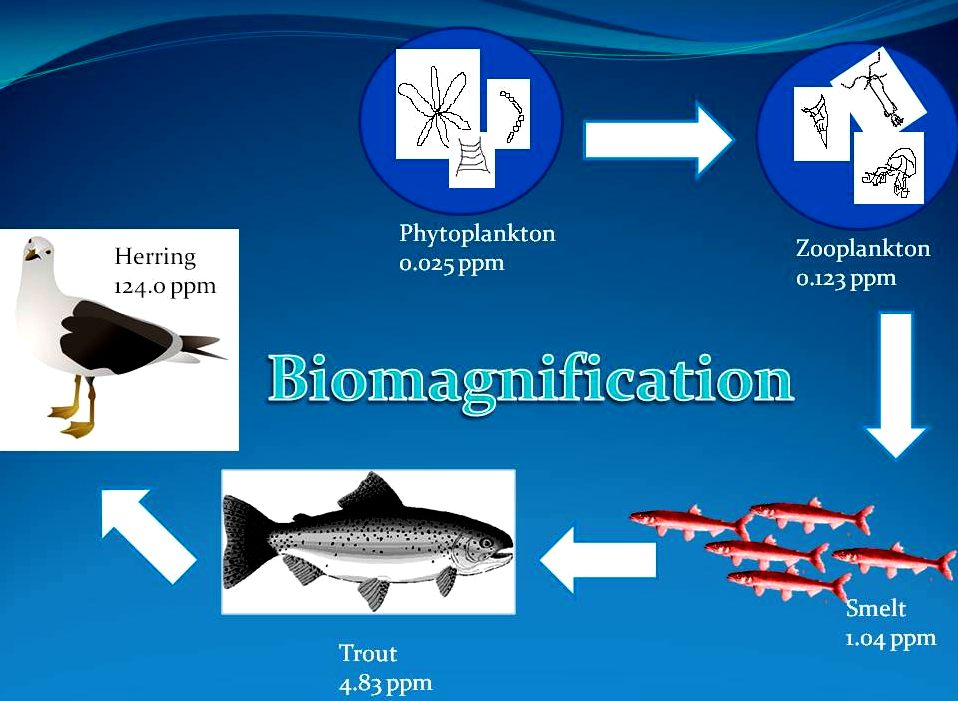
BIOACCUMULATION LEADS TO BIOMAGNIFICATION
As chemicals are accumulated in an organism, it becomes part of the parts that are generally eaten by the next level or
organisms along the food
chain. We mentioned that the energy transferred to the next level is always a fraction of what is available in the previous level. This is because the organisms of the previous level uses energy it has. However, extra nutrients (or chemicals) are of no use to the organisms there. Therefore, all of it is transferred to the next level with a fraction of the previous available energy. In total, we now have less energy and a greater proportion of chemicals in the next level. This process of increase in concentration of chemicals or any toxic substance from one trophic level to the next is called biomagnification.
LINKS
& REFERENCE
https://plasticoceans.org/
http://journals.plos.org/plosone/article?id=10.1371/journal.pone.0191759
https://www.independent.co.uk/environment/plastic-microplastic-microbeads-pollution-toxic-environment-house-of-commons-environmental-audit-a7011256.html
https://www.newsdeeply.com/oceans/articles/2018/04/03/heavy-metal-the-new-toxic-danger-posed-by-ocean-plastic-trash
https://en.wikipedia.org/wiki/Biomagnification
ABS
- BIOMAGNIFICATION
- BP DEEPWATER - CANCER
- CARRIER BAGS
- CLOTHING - COTTON BUDS - DDT - FISHING
NETS
FUKUSHIMA - HEAVY
METALS - MARINE LITTER
- MICROBEADS
- MICRO
PLASTICS - NYLON - OCEAN GYRES
- OCEAN WASTE
PACKAGING - PCBS
-
PET - PLASTIC
- PLASTICS
- POLYCARBONATE
- POLYSTYRENE
- POLYPROPYLENE - POLYTHENE - POPS
PVC - SHOES
- SINGLE USE
- SOUP - STRAWS - WATER
This
website is provided on a free basis as a public information
service. copyright © Cleaner
Oceans Foundation Ltd (COFL) (Company No: 4674774)
2018. Solar
Studios, BN271RF, United Kingdom.
COFL
is a charity without share capital.
|




